Microfluidics
Transport of fluids at field scale and immobilization of nonwetting phases is ultimately determined by pore-scale and fracture-scale processes. Hence, microscopic observations are valuable to interpret larger-scale phenomena (i.e., core scale). Such observations also help us to deepen our mechanistic understandings and provide validation of pore network models and direct numerical simulations of fluid flow.
We create microfluidic devices, referred to as micromodels, using electronics grade silicon wafers and photolithographic techniques. Two-dimensional micromodels allow direct observation of pore-scale events as well as provide understanding of the movement of dispersed bubbles. Micromodels contain an etched pore network pattern that is directly observable with a microscope. A first step is the imaging of representative pore features in rock thin sections. The end result of fabrication is a two-dimensional medium with a precisely known pore network pattern, number of pores, and geometry. To date, we have created micromodels of Berea sandstone, a heavy-oil sand, an idealized rough fracture, a dual porosity limestone, and a meter-long capillary on a chip as shown in the figure below.
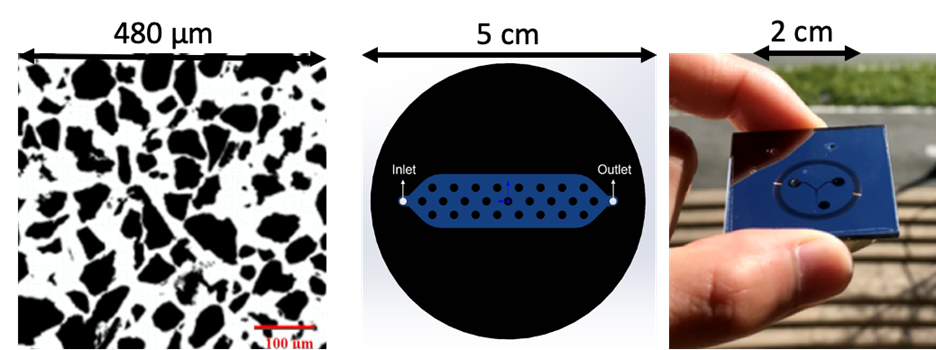
Selected microfluidic designs and devices: (a) grains in black and pore space in white for a weakly consolidated sandstone, (b) 2cm by 7 cm fracture replicated in a micromodel with fracture asperities in black and unobstructed fracture aperture of 25 µm in blue, and (c) a 2 m long capillary tube on a chip. Figures courtesy of Negar Nazari and Wonjin Yun.
We are using microfluidic devices to investigate the mechanics of residual nonwetting phase trapping and the subsequent dissolution of residual phase into the surrounding aqueous phase. This work has application to carbon dioxide storage as well as to water-alternating-gas (WAG) processes. Similarly, we are visualizing (and modeling—see enhanced recovery) the texture (or bubble density) of aqueous wetting foams for mobility control of carbon dioxide in fractures. Conditions span wet to dry foams as well as a wide range of capillary number.
Additionally, we have been successful in the design and manufacture of capillaries with 10’s of µm cross sectional dimensions that are over a meter long. These capillary tubes on a chip are important devices for applications to the dynamics of bubble flow, homogeneous precipitation under flow, and minimum miscibility pressure of carbon dioxide.